Scientific laboratory testing has demonstrated that food and food packaging in the U.S. contains toxic chemicals/additives. Yet the U.S. Food and Drug Administration (FDA) lacks the legal power to compel food manufacturers to provide information to the FDA so they can review the level of public health risks of the additives/chemicals in question (like PFAS and BPA in food packaging that migrates into the food). Now, the U.S. Government Accountability Office (GAO)–our heroes, because they are the only body that keeps the government departments honest–has conducted an investigation and created a report that has recommended that FDA request this authority from Congress.
Here is a quick, at-a-glance summary of the GAO’s report:
.

What GAO Found
The Department of Health and Human Services’ Food and Drug Administration (FDA) has primary responsibility for reviewing the safety of food contact substances before and after they enter the market. These substances come into contact with food during processes such as manufacturing, packaging, and transporting food (see fig.). Such substances may migrate into food, where they could pose a risk to human health. FDA conducts premarket reviews of the safety of substances largely by reviewing companies’ submissions of supporting evidence before substances enter the market. FDA bases its postmarket reviews on safety information, including new information published since the substance’s initial approval for use.
Since 2000, FDA has helped to stop the use of three types of unsafe substances, such as some per- and polyfluoroalkyl substances (PFAS) that are used to greaseproof food packages. These substances may cause health effects, such as liver damage. However, FDA faces two limitations that impede a risk-informed, postmarket review process for food contact substances:
(1) FDA does not have specific legal authority to compel companies to provide information and data on substances’ safety and extent of use. FDA needs such information to prioritize and conduct postmarket reviews. FDA officials said they have begun to develop options to systematically reassess the safety of food additives, which include food contact substances, in response to a House Appropriations Committee report. Also, FDA has a strategic plan to improve data-driven, postmarket surveillance of substances added to the food supply. In its report to the committee, FDA could support its strategic plan and have additional options for obtaining information on food contact substances if it requested specific authority to compel companies to provide information and data on food contact substances’ safety and use.
.
(2) FDA staff can search the agency’s information system for each food contact substance and find the date of the last premarket review. However, FDA’s system cannot readily identify all substances that, according to their last review dates, may warrant additional review because new safety information may have emerged. Tracking review dates in a way that allows FDA to identify these substances may help address challenges FDA faces in making risk-informed decisions on which substances to prioritize for postmarket review.
–
Why GAO Did This Study
Thousands of food contact substances are available for use in manufacturing, packaging, and transporting food. These substances are used, for example, in food wrappers and in the lining of metal food cans. Academic, consumer, and other stakeholders have raised concerns that some of the substances may, by themselves or in combination with other substances, contribute to adverse health effects, such as thyroid disease and hormone disruption.
This report (1) identifies the primary means that companies use to bring food contact substances to the market and describes FDA’s premarket safety review process and (2) examines FDA’s postmarket safety review actions and the limitations of such reviews. To conduct this work, GAO interviewed FDA officials and stakeholders and reviewed, among other things, FDA documents and the agency’s website for actions it took from January 2000 to May 2022 to stop the use of potentially unsafe substances.
.
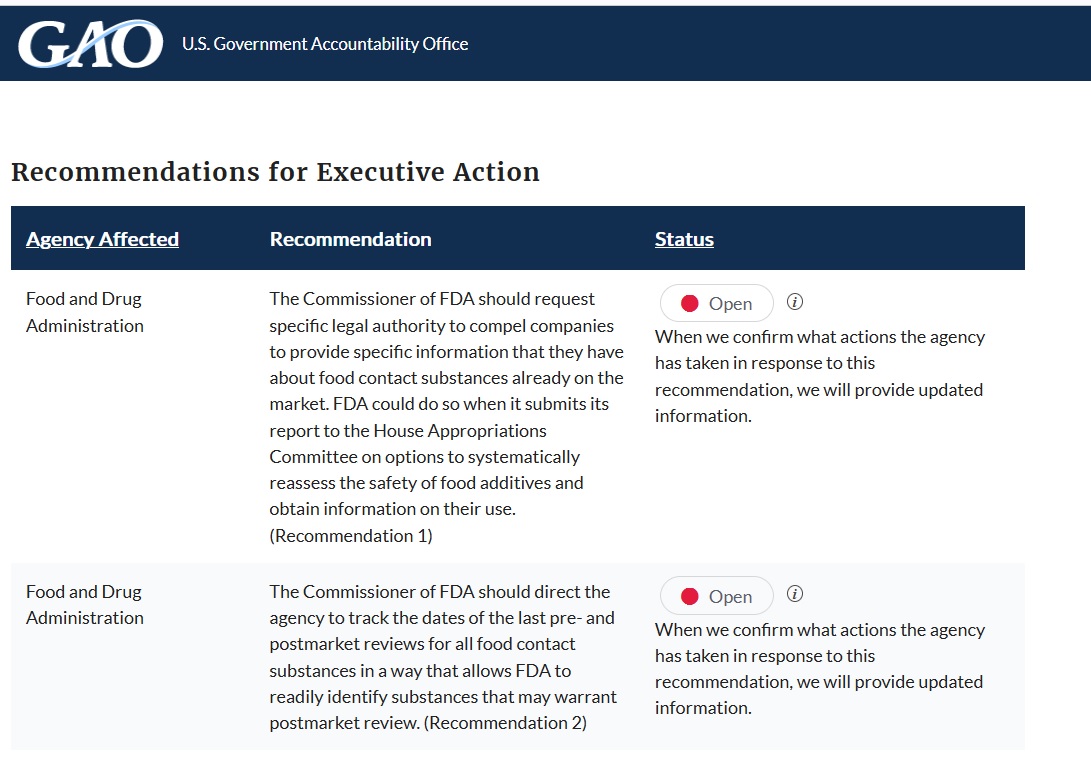
Recommendations
GAO is making two recommendations to FDA to (1) request from Congress specific legal authority to compel companies to provide the information needed to reassess the safety of substances and (2) track the dates of the last reviews for all food contact substances to allow FDA to readily identify substances that may warrant postmarket review. FDA neither agreed nor disagreed with the first recommendation and agreed with the second recommendation.
–
–


